Experiment designA recent study by Yi and his colleagues has shown the potentials of non-depleting anti-CD4 and anti-CD8 antibodies in the induction of type I diabetes remission. The study has demonstrated rapid remission of type I diabetes in non-obese diabetic (NOD) mice treated with YTS 105 (anti-CD8 antibody) and YTS 177 (anti-CD4 antibody). To do this, the study utilised female NOD mice with blood glucose levels > 250mg/dL and administered them with intraperitoneal injections of 600-800μg of YTS177 and YTS105 or rat IgG2a isotype control monoclonal antibody (mAb). No exogenous insulin was provided to these mice prior to antibody treatment and at the time of the treatment.
Results1. Rapid and long-term remission
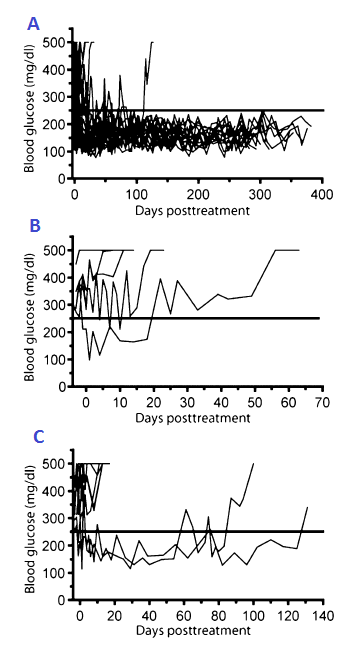
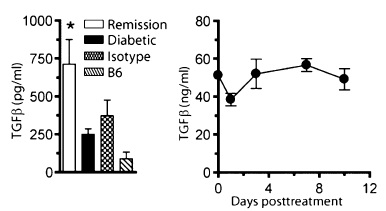
In recent-onset diabetic NOD mice, diabetes was reversed in 79% of YTS-treated mice (19 out of 24 mice) while there was no remission evident in the control group. Remission was shown in 54% of mice (13 out of 24) within 72 hours of treatment with average blood glucose level from 365mg/dL to 231mg/dL (Figure 1). Interestingly, only the mice that were treated with YTS177 and YTS105 together went into remission while the mice that were treated with YTS177 or YTS105 alone did not go into remission or went into transient remission (Figure 2). After treatment, 18 out of 19 of remission NOD mice remained in its recovery state for over 200 days with some of them remained euglycemic up to 400 days in the absence of exogenous insulin. The mice which have undergone remission showed smaller islet regions indicative of the absence of inflammation in comparison to diabetic-remaining mice with 40% of their islets exhibiting signs of inflammation. Prompt effects of YTS treatment is desirable due to limited time to rescue adequate numbers of beta-cells following the onset of the disease. 2. Purging of the pancreatic and pancreatic lymph node (PLN) T-cells There were significant reductions in pancreatic interferon-γ (IFN-γ) and interleukin-2 (IL-2) in NOD mice in their remission state demonstrating the protective effects of YTS treatment. Also, the numbers of pancreatic CD4+ and CD8+ cells were significantly reduced approximately three to fivefold 6 days after the first YTS injection (Figure 3). The reduction in T-cell numbers in the pancreas was independent of T-cell subpopulations. CD4+ cells in PLN were decreased by two folds while CD8+ cells were slightly decreased. On the other hand, an increase in the number of CD4+ and CD8+ cells in the spleen were observed. 3. T-cell clearance mechanism YTS treatment is independent of apoptosis. The non-depleting nature of YTS 177 and YTS 105 resulted in no changes in the systemic T-cell numbers in NOD mice. This aspect is an advantage of YTS treatment compared to previous studies of anti-CD3 antibodies that are associated with T-cells apoptosis which may result in a compromised normal protective immunity. YTS treatment works by binding to the T-cells and decreasing the retention cues present during inflammation in the pancreas and PLN. In other words, YTS bound T-cells lose their ability to respond to retention signals in the pancreas and PLN, and this promotes rapid T-cell purging of the pancreas (and PLN) in a tissue-specific manner. In addition, another possible non-apoptotic mechanisms such as T-cell redistributions and/or cytotoxic cell deaths could play a role in the tissue-specific T-cell purging. 4. Transforming growth factor-β1 (TGF-β1) is required for the induction of YTS177- and YTS105-mediated remission The level of TGF-β1 in pancreas increased by more than two folds in remission NOD mice compared to diabetic-remaining NOD mice and nonautoimmune control mice (Figure 4). In contrast, serum TGF-β did not increase in remission NOD mice. TGF-β1 but not IL-10 were necessary for inducing remission verified after injections of 200ug of anti-TGF-β and 300ug anti-IL-10 receptor 3 times per week for 3 weeks. It was found that the source of these TGF-β cells originated from antigen presenting cell (APC) but not from T-cells. Also TGF-β1 cells do not target T-cells and is regulatory T-cell (Treg) independent. The mechanism of how TGF-β affects YTS treatment is uncertain, however, it is hypothesised that they might inhibit the actions of pancreatic APC or directly increase the survivability of pancreatic β-cells. 5. Factors maintaining the effect of remission Although induction of remission is independent of Treg, maintenance of remission appeared to be associated with β-cell specific Treg and enhanced by Foxp3+ Treg originated in the PLN. Furthermore, there was a significant decrease in T-cell reactivity to insulin B chain in the PLN of remission mice. Expansion of Treg was enabled by purging of T-cells in the PLN (and pancreas) by YTS treatment. |
|
Advantages
YTS treatment is antigen specific
This experiment was highly specific to pancreatic CD4+ and CD8+ T-cells which damages beta-cells in the pancreas. Due to the inactivation and loss of T-cells in pancreas, there were increase in T-cell population in spleen. However the circulating T-cell level in the blood remained the same.
Regulation of T-cells are independent to apoptosis
Unlike some previous immunotherapies such as NFB anti-CD3 and antithymocyteglobulin, targeting YTS treatment does not undergo apoptosis. This aspect makes this treatment less toxic and more reversible, as well as causing less side effects.
This experiment was highly specific to pancreatic CD4+ and CD8+ T-cells which damages beta-cells in the pancreas. Due to the inactivation and loss of T-cells in pancreas, there were increase in T-cell population in spleen. However the circulating T-cell level in the blood remained the same.
Regulation of T-cells are independent to apoptosis
Unlike some previous immunotherapies such as NFB anti-CD3 and antithymocyteglobulin, targeting YTS treatment does not undergo apoptosis. This aspect makes this treatment less toxic and more reversible, as well as causing less side effects.
Limitation
Delayed YTS treatment or NOD mice with blood glucose levels >400mg/dL failed
YTS treatment is only limited to 'early-onset' diabetes because this treatment is independent to insulin. Therefore normal functioning pancreatic beta-cells are required to produce insulin.
YTS treatment is only limited to 'early-onset' diabetes because this treatment is independent to insulin. Therefore normal functioning pancreatic beta-cells are required to produce insulin.
T.N. Budi, A.N. Khuong, C. YeoJoon | Copyright ©2012